Since calculations using PM3 are much faster than by DFT, and since this semi-empirical method supplies good estimates of the equilibrium geometries, all systems were first optimised using PM3 and then the final values for the energies were obtained by DFT. All DFT calculation were done using the non-local BP86 functional with DN** numerical polarization basis set. Halogen periodic table pro#Ramôa Ribeiro, in Studies in Surface Science and Catalysis, 2001 2 COMPUTATIONAL METHODSĪll computations were carried out using either PC Spartan Plus or PC Spartan Pro (from Wavefunction inc.) and both DFT and a semi-empirical method, PM3, were used. However, due to issues pertaining to quantity and cost of the reagent, the method has largely failed to encourage scientists and process engineers to implement this technique in industry. The leaching process offered by iodide is rapid, noncorrosive, nontoxic, and highly efficient in terms of chemical selectivity. On the other hand, iodide leaching represents a promising technology as compared to chloride method. Since the reaction scheme pertaining to halides of chlorine is thermodynamically favored by the presence of an oxidizing agent, the results with sodium chlorate (NaClO 3), rich in oxygen content, proved to be the best lixiviant for the leaching of gold, although the leaching rate is below 60%. demonstrated via experiments to determine the efficiency of various chlorine-based compounds (Cl 2, NaClO, NaClO 3). Several studies have been carried out to find out the effectiveness of chlorine-based halogens. Among various halogen compounds, chlorine/chloride-based treatment methods are widely followed in industries. Several studies conducted by the researchers reveal that the leaching method induced by bromine lixiviant is less often used as the vapor pressure of bromine is low (28 KPa at 35☌) and needs specialized equipment to comply with safety and health regulations. Later, a similar method has been tested by many researchers to recover pure metals from PCB wastes. Halogenation method is traditionally employed to recover metals, especially Au, from its ores. Where M, L 2, and L − represent precious metals, a halogen element, and the oxidizing agent, respectively. The complex structures of these materials have been the subject of considerable research. These are really mixed valence 3+, 2+ compounds with formulas such as Dy 5Cl 11, Yb 6Cl 13, and Sm 11Br 24. For example, discreet compounds with halogen to rare earth ratios of 2:200, 2:167, and 2:140 have been observed for chlorides and bromides of Dy, Yb, Ho, and Sm. In addition to the above “normal” valence materials, recent research has uncovered a number of mixed valence and apparent subvalent halides. The compounds formed when RE = La, Ce, Pr, and Gd all have high electrical conductivities and must be regarded as trivalent as with the hydrides. Halogen periodic table series#The most extensive series of REX 2 compounds occurs for X = I which numbers 11 at present. For X = F, the only REX 2 that exist have RE = Sm, Eu, and Yb. Not surprisingly, REX 3 compounds exist for all RE and all X. The REX 4 compounds are least common, existing only for X = F and RE = Ce, Pr, and Tb, those commonly exhibiting a 4+ valence. If we represent a “halide” ion as X −, compounds of composition REX 4, REX 3, and REX 2 form readily and have been known for some time. The rare earths lose their valence electrons, donating them to the halogens to form halide ions F − (fluoride), Cl − (chloride), Br − (bromide), and I − (iodide). The behavior of the rare earths with the halogen elements fluorine (F), chlorine (Cl), bromine (Br), and iodine (I) is somewhat similar to that with hydrogen. 
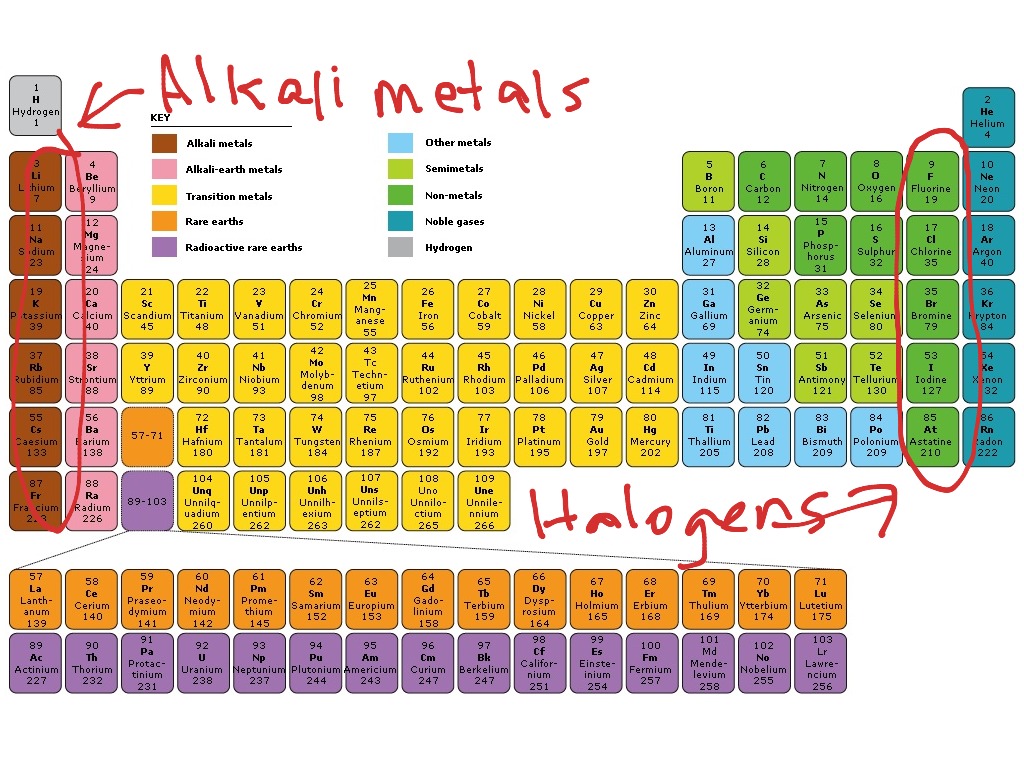
Greedan, in Encyclopedia of Physical Science and Technology (Third Edition), 2003 III.A.4 Halides (Fluorides, Chlorides, Bromides, and Iodides) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
